
In the case of acetic acid, the R represents the methyl group. The below image shows a carboxyl group where R represents the rest of the molecule to which the carboxyl group is attached the letter R is sometimes replaced by a wiggly line. In the case of acetic acid, the free electron binds with a carboxyl group (CO 2H, -COOH or -C(=O)OH) which is a single carbon atom bonded to a hydroxyl group (-OH) and double-bonded to an oxygen atom. The picture shows a methane molecule composed of a methyl group and an extra hydrogen atom. With a free electron, methane reacts with ozone (O 3) to produce carbon dioxide and water in the following reaction: (3)CH 4 + (4)O 3 = (3)CO 2 + (6)H 2O. The simplest carbon molecule is methane (CH 4), well known for its contribution to global warming. 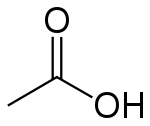
As carbon has four electrons, the free electron usually bonds with other molecules by way of a covalent bond. They are composed of three hydrogen atoms and one carbon atom (CH 3). Methyl groups are one of the most common organic compounds on the planet but are rarely found as single entities. The acetic acid formula is a simple one and the result of a methyl group and a carboxyl group. When water is added, these bonds are broken and the crystalline form dissolves. In crystalline form, two acetic acid molecules join together with hydrogen bonds to form a dimer. 
More information pertaining to acetic acid uses will be discussed later on in this article. This means it has many uses in industrial chemical production but has also gained a reputation as a weight-loss supplement as it affects fat and sugar metabolism. Acetic acid dissociationĪs a solvent, liquid acetic acid dissolves polar (hydrophilic) compounds such as salts and sugars and non-polar compounds which include fats and oils. We say that the water molecule is protonated or has had a proton (hydron) donated to it. Acetic acid passes on a hydrogen ion to the water molecule to produce a hydronium ion. To the left are a single acetic acid molecule and a single water molecule. The following image shows the dissociation of acetic acid to acetate in water. Even glacial acetic acid has a small quantity of water. 
A solution always contains water even modern superacids such as carborane are dissolved in concentrated aqueous solutions of other acids. A pH value is also only given to a solution. The pH of a solution is, therefore, dependent upon the balance of hydronium and hydroxyl and not the number of hydrogen ions, although these will affect this balance. This means there are more hydronium ions and, therefore, create a positively charged (or acidic) solution. A small percentage of positively charged hydrogen ions bind to the water molecules and turn them into H 30 +. It is, therefore, possible to understand the alternative name of acetic acid – hydrogen acetate. When acetic acid is added to water, it splits into a negatively charged acetate ion (CH 3COO –) and H +. Two molecules of water (H 20) are formed when a hydronium and hydroxyl ion bind and the positive and negative charges are canceled out. Neutral solutions (neither acid nor alkaline) contain a balanced number of hydronium ions (H 30 +) and hydroxyl ions (OH –). It should be made clear that it is not the presence of a single hydrogen atom that changes the pH of a solution. In comparison with the world’s strongest acid – carborane acid (H(CHB 11Cl 11)) – with a pH value of -18, acetic acid is mildly acidic in comparison. Vinegar is a solution of acetic acid and water where approximately 0.4% of acetic acid molecules give up their H + atoms leading to an acidic solution of approximately 2.4 pH. The donated hydrogen is dissociated from the carboxyl group. This characteristic means it is a member of the Brønsted acid group where protons are donated to acceptor molecules known as Brønsted bases. Acetic acid or ethanoic acid is a protic solvent it is able to donate protons in the form of hydrons (positively charged hydrogen atoms). Vinegar: an acetic acid solution Acetic Acid StructureĪcetic acid structure is that of a simple carboxylic acid and consists of a methyl group attached to a carboxyl group as seen in the image below. The molecular formula of acetic acid is C 2H 4O 2 or CH 3COOH, where –COOH defines the presence of the single carboxyl group. Otherwise known as ethanoic acid, methanecarboxylic acid, hydrogen acetate or ethylic acid, this organic compound is used in chemical manufacturing, as a food additive, and in petroleum production. Acetic acid is a mildly corrosive monocarboxylic acid.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
